A novel Raman chemical sensor made from noodlelike threads of gold - ScienceDaily. 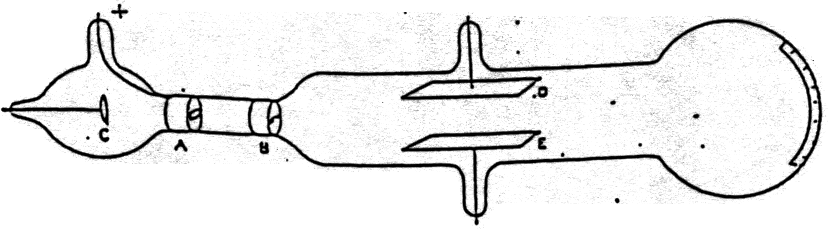
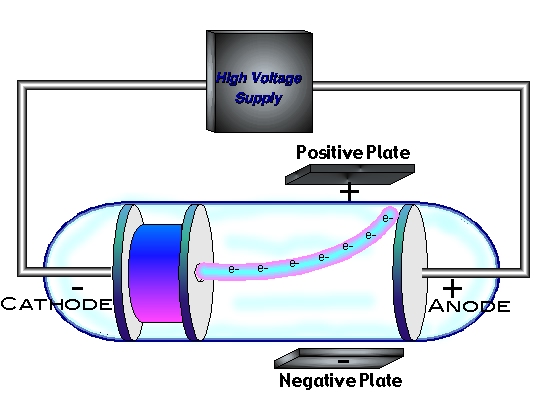
Physical and Chemical Properties of Carboxylic acids.Kohlrausch law of independent migration of ions.Organocatalytic Synthesis of Thiiranes from Alkenes.The study of the atom as well as its structure has paved the way for numerous other inventions that have played a significant role in the development of mankind. Thomson’s model proved to be the base for the development of other atomic models even though it is not an accurate model to account for the atomic structure. These is no experimental evidence in its support.Thomson’s model failed to explain how thin metal foils can cause the scattering of alpha particles.Therefore, this theory also failed to tell us about the position of the nucleus in an atom It failed to explain the stability of an atom because his model of atom failed to explain how a positive charge holds the negatively charged electrons in an atom.They were the first ones to demonstrate it. Thomson also demonstrated the ionization of air by X-ray along with Rutherford. It must be noted here that in this model the mass of the atom is assumed to be uniformly distributed over the atom. This makes the atom as a whole to be electrically neutral. Both the negative and positive charges are equal in magnitude.An atom consists of a positively charged sphere with electrons embedded into it.Thomson was successful is explaining the overall neutrality of an atom. In his atomic structure model, he considered atoms to be surrounded by a cloud having positive as well as negative charges. He believed that an atom is made up of thousands of electrons and assumed that an electron is two thousand times lighter than a proton. We may also think link this to a watermelon, in which the positive charge in the atom is spread all over like the red edible part of the watermelon, while the electrons are embedded in the positively charged sphere, exactly like the seeds in the watermelon. In a sphere of positive charge, the electrons were like currants (dry fruits) in a spherical Christmas pudding. The model of an atom proposed by Thomson was compared with a Christmas pudding by him. This experiment took place in the year 1897. The negative particle was called an electron. Thomson during cathode ray tube experiment. a negatively charged particle was discovered by J.J. For his work on the discovery of electrons he was awarded the Nobel Prize in Physics in 1906. Thomson was a British physicist, and was born in Cheetham Hill, a suburb of Manchester, on 18 December 1856. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
